First, you must know that doing research with Cannabis products is virtually impossible in the USA today. The effect of all the governmental legal restrictions, virtually stops all research. We were most fortunate to find a wonderfully supportive group of lactation consultants and cannabis using moms in Colorado, who helped us find breastfeeding mothers who were using this product while breastfeeding. Nine of these moms followed our directions, and provided milk samples. This is one of the first really accurate assessments of THC transfer into human milk. What it suggests to us is that THC does transfer into human milk, and it is apparetly a function of the daily use in mothers. We think that in occassional users, the level of transfer is minimal (about 2.5% or lower). We also surmise, that in heavy users it could be 3 times this dose. This is probably a function of the filling of the adipose sites with chronic daily use. Once these fat sites are FULL of THC, less is transferred out to fat tissues following smoking...thus leaving more in the plasma compartment to transfer into milk. We strongly urge cannabis using moms to avoid using these products while breastfeeding, as we simply do not know what happens neurobehaviourally to these exposed infants. Again, thanks to those wonderfull support groups that helped Dr. Baker and I do this beginning study. TWH
Title: Transfer of Inhaled Cannabis Into Human Breast Milk.
Authors: Baker T, Datta P, Rewers-Felkins K, Thompson H, Kallem RR, Hale TW.
Obstet Gynecol. 2018 May;131(5):783-788
Abstract:
To evaluate the transfer of delta-9-tetrahydrocannabinol and its metabolites into human breast milk after maternal inhalation of 0.1 g cannabis containing 23.18% delta-9-tetrahydrocannabinol. In this pilot pharmacokinetic study, breast milk samples were collected from mothers who regularly consumed cannabis, were 2-5 months postpartum, and exclusively breastfeeding their infants. Women were anonymously recruited for the study. After discontinuing cannabis for at least 24 hours, they were directed to obtain a baseline breast milk sample, then smoke a preweighed, analyzed, standardized strain of cannabis from one preselected dispensary, and collect breast milk samples at specific time points: 20 minutes and 1, 2, and 4 hours. Quantification of delta-9-tetrahydrocannabinol and its metabolites in these collected breast milk samples was performed by high-performance liquid chromatography tandem mass spectrometry. A total of eight women were enrolled. Most were occasional cannabis smokers and one a chronic user. Delta-9-tetrahydrocannabinol was detected at low concentrations at all the time points beyond time zero. No metabolites were detected at any time point. Delta-9-tetrahydrocannabinol was transferred into mother's milk such that exclusively breastfeeding infants ingested an estimated mean of 2.5% of the maternal dose (the calculated relative infant dose=2.5%, range 0.4-8.7%). The estimated daily infant dose was 8 micrograms per kilogram per day. This study documents inhaled delta-9-tetrahydrocannabinol transfer into the mother's breast milk. Low concentrations of delta-9-tetrahydrocannabinol were detected. The long-term neurobehavioral effect of exposure to delta-9-tetrahydrocannabinol on the developing brain is unclear. Mothers should be cautious using cannabis during pregnancy and breastfeeding.
Table: Pharmacokinetic Parameters of delta-9-THC in human milk samples (n=9).
Parameter (units) Value
AUC (ng.h/mL) 208.2
Cavg (ng/mL) 52.05
Cmax (ng/mL) 88.21
Tmax (h) 1
Infant dose (μg/kg/day) 7.8
RID (%) 2.4
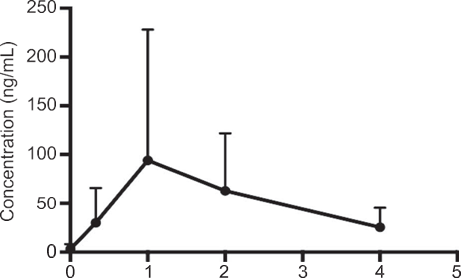
Figure: Mean concentration time profile of delta-9-tetrahy-drocannabinol in human breast milk (mean± SD, n=8).
PMID29630019
